
Some sources follow a compromise that puts La–Lu and Ac–Lr as the f-block rows (despite that giving 15 f-block elements in each row, which contradicts quantum mechanics), leaving the heavier members of group 3 ambiguous. Arguments can still occasionally be encountered in the contemporary literature purporting to defend it, but most authors consider them logically inconsistent. General inorganic chemistry texts often put scandium (Sc), yttrium (Y), lanthanum (La), and actinium (Ac) in group 3, so that Ce–Lu and Th–Lr become the f-block between groups 3 and 4 this was based on incorrectly measured electron configurations from history, and Lev Landau and Evgeny Lifshitz already considered it incorrect in 1948. This makes the group somewhat exceptional.ī The 14 f-block groups (columns) do not have a group number.Ĭ The correct composition of group 3 is scandium (Sc), yttrium (Y), lutetium (Lu), and lawrencium (Lr), as shown here: this is endorsed by 19 IUPAC reports on the question. Hydrogen is not considered to be an alkali metal as it is not a metal, though it is more analogous to them than any other group. Elements of the group have one s-electron in the outer electron shell. Ī Group 1 is composed of hydrogen (H) and the alkali metals. In history, several sets of group names have been used, based on Roman numberings I–VIII, and "A" and "B" suffixes. Also, trivial names (like halogens) are common. Modern group names are numbers 1–18, with the 14 f-block columns remaining unnumbered (together making the 32 columns in the periodic table). In astrophysics and nuclear physics, it usually refers to iron, cobalt, nickel, chromium, and manganese. An exception is the " iron group", which usually refers to " group 8", but in chemistry may also mean iron, cobalt, and nickel, or some other set of elements with similar chemical properties. For example, group 16 is also described as the "oxygen group" and as the " chalcogens". Groups may also be identified using their topmost element, or have a specific name. Similar variation on the inner transition metals continues to exist in textbooks, although the correct positioning has been known since 1948 and was twice endorsed by IUPAC in 1988 (together with the 1–18 numbering) and 2021. 
The system of eighteen groups is generally accepted by the chemistry community, but some dissent exists about membership of elements number 1 and 2 ( hydrogen and helium). It replaces two older incompatible naming schemes, used by the Chemical Abstract Service (CAS, more popular in the United States), and by IUPAC before 1988 (more popular in Europe). The modern numbering system of "group 1" to "group 18" has been recommended by the International Union of Pure and Applied Chemistry (IUPAC) since 1988. There are three systems of group numbering for the groups the same number may be assigned to different groups depending on the system being used. The elements in a group have similar physical or chemical characteristics of the outermost electron shells of their atoms (i.e., the same core charge), because most chemical properties are dominated by the orbital location of the outermost electron. There are 18 numbered groups in the periodic table the 14 f-block columns, between groups 2 and 3, are not numbered. In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. In the periodic table of the elements, each column is a group. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Column of elements in the periodic table of the chemical elements Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). 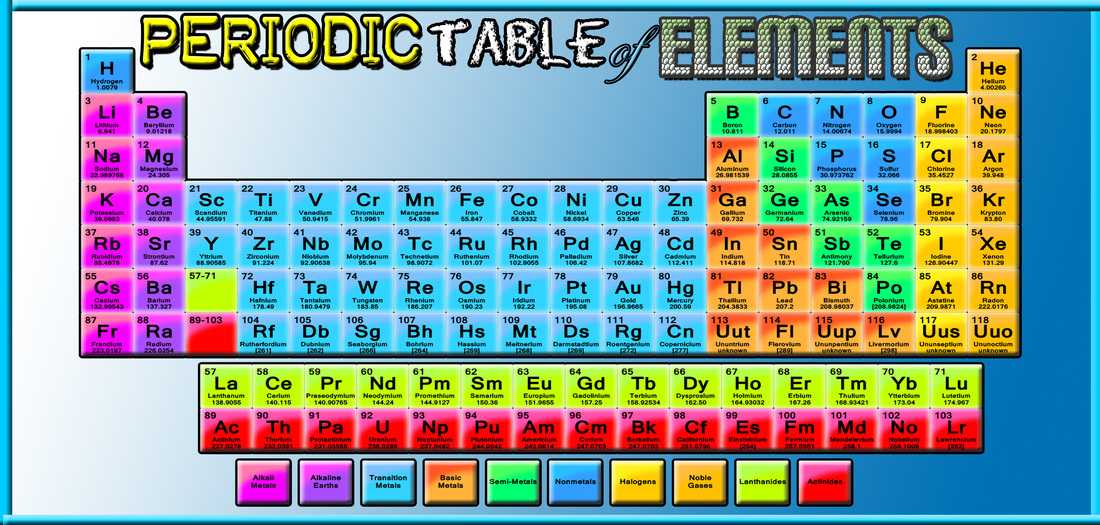
Free Gift for you: Interactive Periodic Table In this way, the elements of the same group show similar chemical properties and they also have the same number of valence electrons. They are soft and can be cut easily with a kitchen knife.Īlso all the elements of group 1 have one valence electron.Īll the elements of group 18 are chemically inert (that means they do not easily react with other elements).Īnd all the elements of group 18 have a complete octet (that means they have 8 electrons in their outer shell). 
The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons.Īll the elements of group 1 are highly reactive to water. There are total 18 vertical columns on periodic table. Groups are the vertical columns on the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
